Hand-washing: The chemistry of soap against the deadly Covid-19
- Hand-washing with soap and water for 20 seconds has been recommended to combat the threat of the coronavirus outbreak.
- But many people still disbelieve this simple precautionary measure to save you from infection and its complications.
- It is simple science which is explained below.
The structure of Covid-19
Virus expert Professor Kurt Krause from the University of Otago states:
“A virus is a very intriguing package of nucleic acid that is wrapped up in a protein exterior and then, in some cases, surrounded by a lipid – or a fat – envelope.”

The lipid cover maintains viral integrity. But this is where soap acts as well. Hence it is a strength as well as a weakness for the virus.
Hand-washing: Soap and water and their chemistry
Water and fat do not mix readily because water is a polar molecule (charged) whereas fat is a non-polar molecule. Water has one end of the molecule slightly positively charged and the other end is negatively charged. And in case of fat, the charge is evenly spread.
Polar molecules like polar molecules and dissolve with each other. But non-polar molecules do not mix with polar molecules.
The basic recipe and ingredients to manufacture a soap has remained the same over the last 5000 years. Fat or oil is mixed with an alkaline substance such as sodium hydroxide or lye.
takes place and the result is a soap. Soap is amphiphilic which means that it likes both water as well as fat. Allan Blackman, Professor at Auckland University of Technology says:
“If you make a molecule that’s got a polar end and a nonpolar end, the polar end really wants to be in the water and the nonpolar end really wants to be in the grease.”
“What really is going on there is [the soap molecule] drags the grease into the water, it kind of makes the grease soluble in water.”

Click to read on Coronavirus danger looms high! How to make your own hand sanitizer!?
Hand-washing: How does soap destroy the virus?
Water aline will not destroy the virus. But when soap is added, the fatty envelope of the virus is destroyed by it. Professor Kurt explains:
“It’s one of those viruses that a good scrubbing with soap and water does quite a good job in breaking down its composition,”
Therefore, a good hand wash with soap and water lasting for twenty seconds is effective in not only washing away the virus but also killing it.
Detergents are similarly effective in eradicating the virus. Antimicrobial soaps are equally effective and not better.
Steps of proper hand-washing
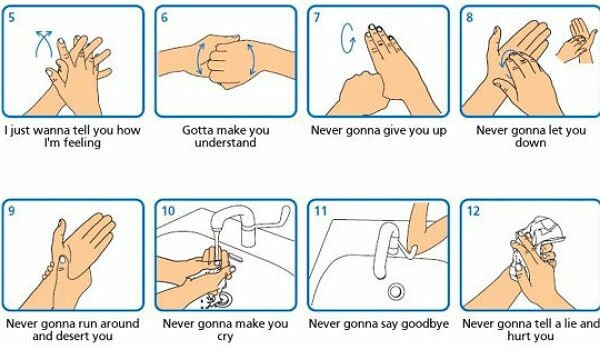
Wash your hands thoroughly at regular intervals to protect yourself and your family against sickness. Hand-washing is mandatory after:
-Before touching or preparing food
-Before eating food
-And Before caring for a sick person
-After touching infected vomitus or stools or treating a wound.
-After visiting the toilet.
-Also, After blowing the nose, sneezing, or coughing.
-After touching garbage or any infected matter.
Wet your hands with clean, running water (warm or cold), then turn off the tap and apply soap. Rub your hands together and make lather with the soap. Rub your hands, between your fingers, and under your nails. Do so for at least 20 seconds.
Rinse clean with water. Air dry or dry with a clean towel or tissue.
Also read Martha MacCallum: questions role of covid vaccine in causing cardiac arrest in Bronny James!
Source: RNZ

